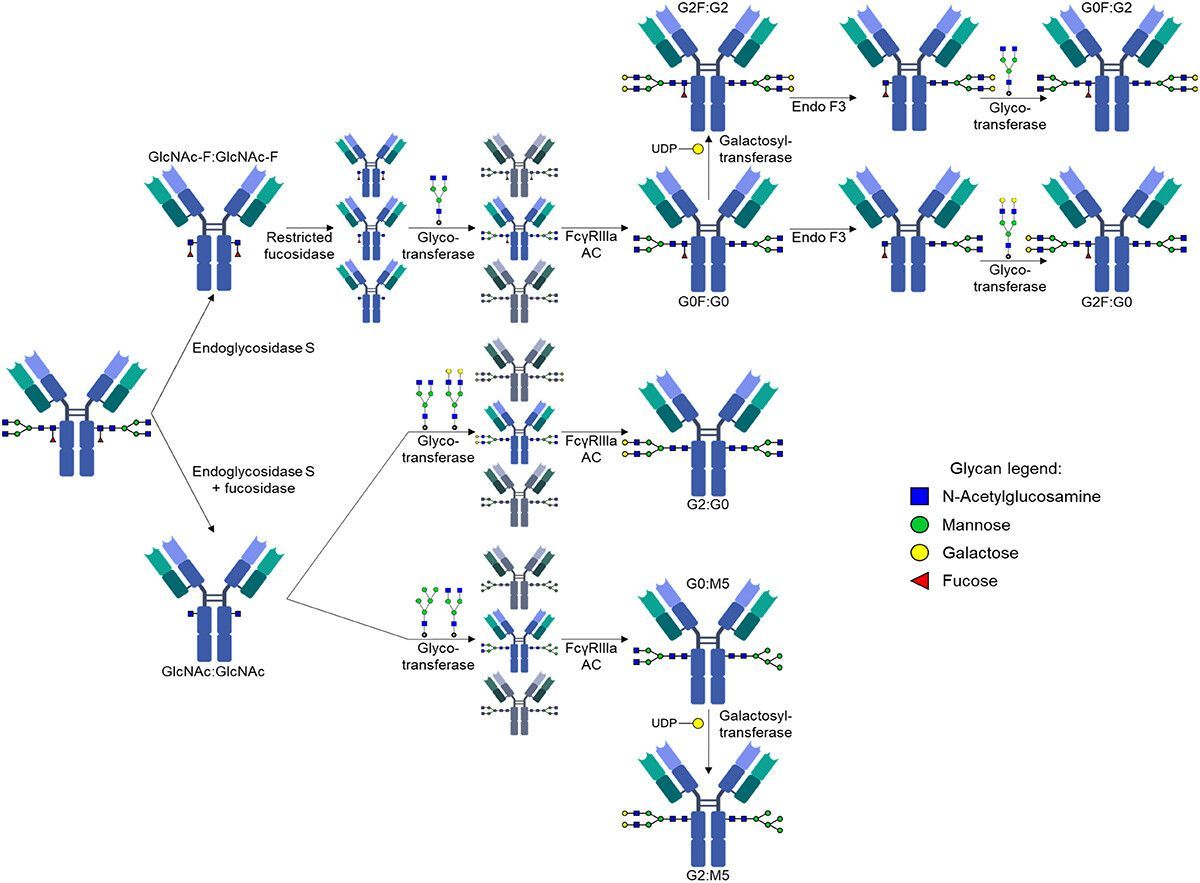
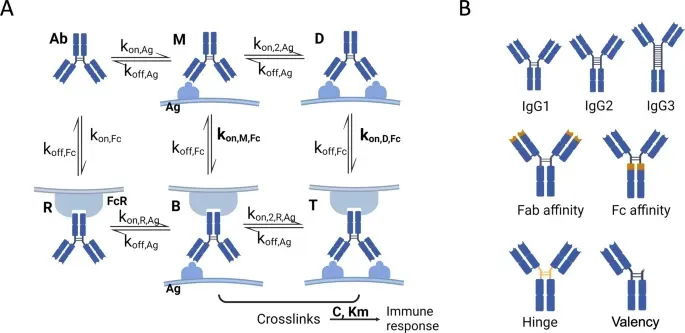
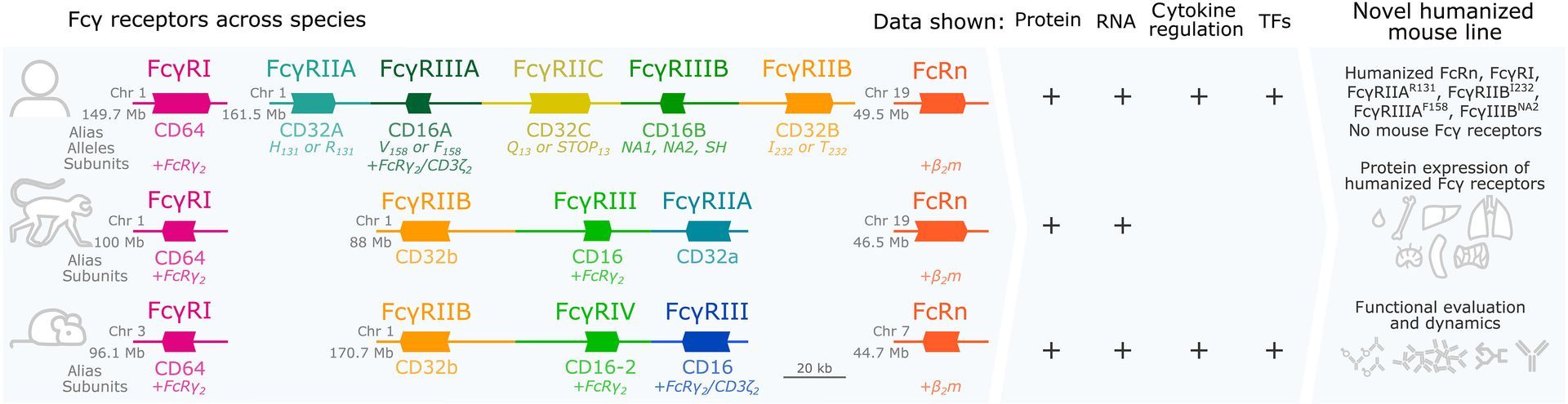
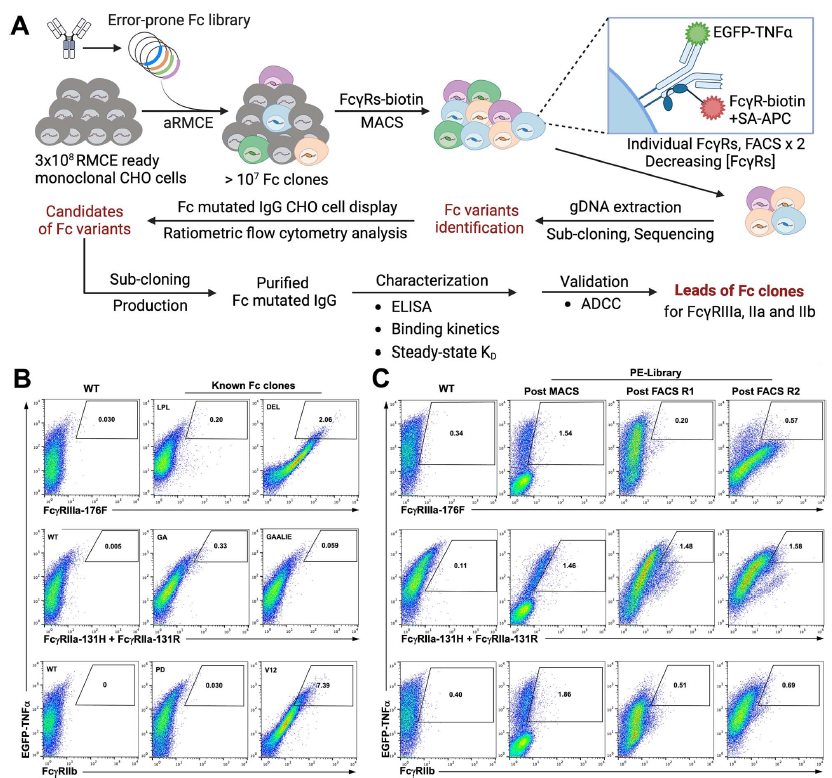
SeromYx Systems, Inc. Appoints Andrew Ball, PhD, as Chief Executive Officer
Woburn, MA, February 5th, 2025 — SeromYx Systems, Inc. , a global leader in high-throughput antibody characterization and Systems Serology services, today announced the retirement of Chief Executive Officer, Mr. Piers Whitehead, a co-founder of the company, who will remain with the organization as a member of the Board of Directors and as a strategic advisor. Concurrently, as part of a planned transition, Andrew Ball, PhD has been appointed Chief Executive Officer and Director, effective February 4th, 2025.
Dr. Ball brings over 20 years of global leadership experience in the life sciences industry, with a successful track record in technical and commercial development of assay platforms and services. Most recently, he served as Vice President, Assay Development Platforms at Abcam, a Danaher company, where he led a global R&D organization developing and commercializing immunoassays, cell-based assays, antibody conjugates, recombinant proteins and contract assay services. He has also held roles as VP of Assay Development at Quanterix, developing ultrasensitive assays for neurodegenerative disease and immunooncology; as VP of R&D at Quad Technologies, a cell and gene therapy startup acquired by Bio-Techne, and has served in a variety of innovation and strategic leadership roles at MilliporeSigma.
Commenting on the appointment, Armin Spura, PhD, Executive Chairman of the Board of Directors said:
“On behalf of the Board, I want to thank Piers for his many contributions to SeromYx’s success since its founding. Under Piers’ leadership, SeromYx has grown to become successful and profitable, powered by the leading technology platform for Fc effector profiling. I am very pleased to announce the appointment of Dr. Andrew Ball as our new Chief Executive Officer. Andrew is a distinguished industry leader whose deep experience in assay development, insights into antibody development, and ability to bridge technical and commercial applications make him the ideal choice to lead SeromYx. We are confident that Andrew’s vision and strategic acumen will position SeromYx for continued success in the years ahead.”
Andrew Ball added:
“ I am excited to join SeromYx at this pivotal time as we continue to deliver for our clients and build on the strong foundations the company has established – with our comprehensive Fc effector function profiling platform. I believe SeromYx is uniquely positioned to deliver new insights into the correlation between function and clinical outcomes during therapeutic antibody discovery and development. “
The company extends its gratitude to Piers Whitehead for his leadership and contributions, which have positioned SeromYx for continued success.
About SeromYx Systems, Inc.:
SeromYx Systems, Inc. is a global leader in providing GCLP high-throughput antibody characterization and Systems Serology services, offering advanced solutions for the comprehensive analysis of immune responses and correlation of antibody function with clinical outcomes. With a dedication to innovation and excellence, SeromYx is at the forefront of revolutionizing the field of vaccine and therapeutic antibody development through its cutting-edge technologies and strategic collaborations.
Contact:
Abigail Harris
Business Development Manager
Abigail.harris@seromyx.com