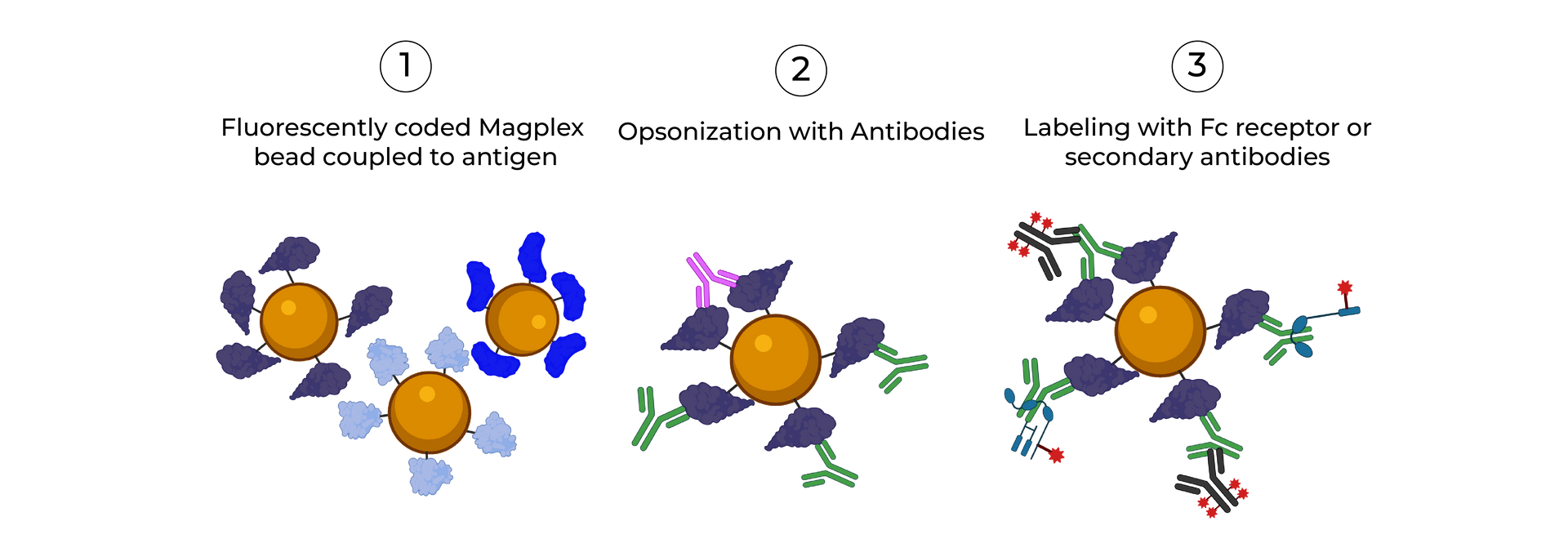
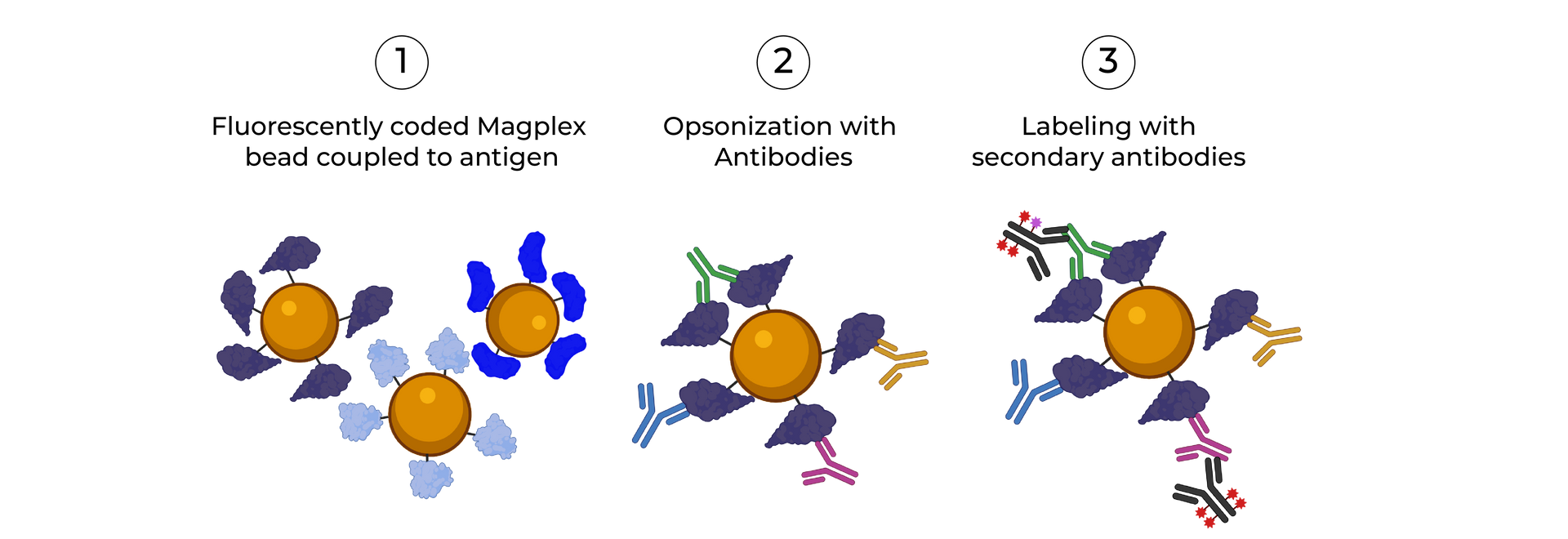
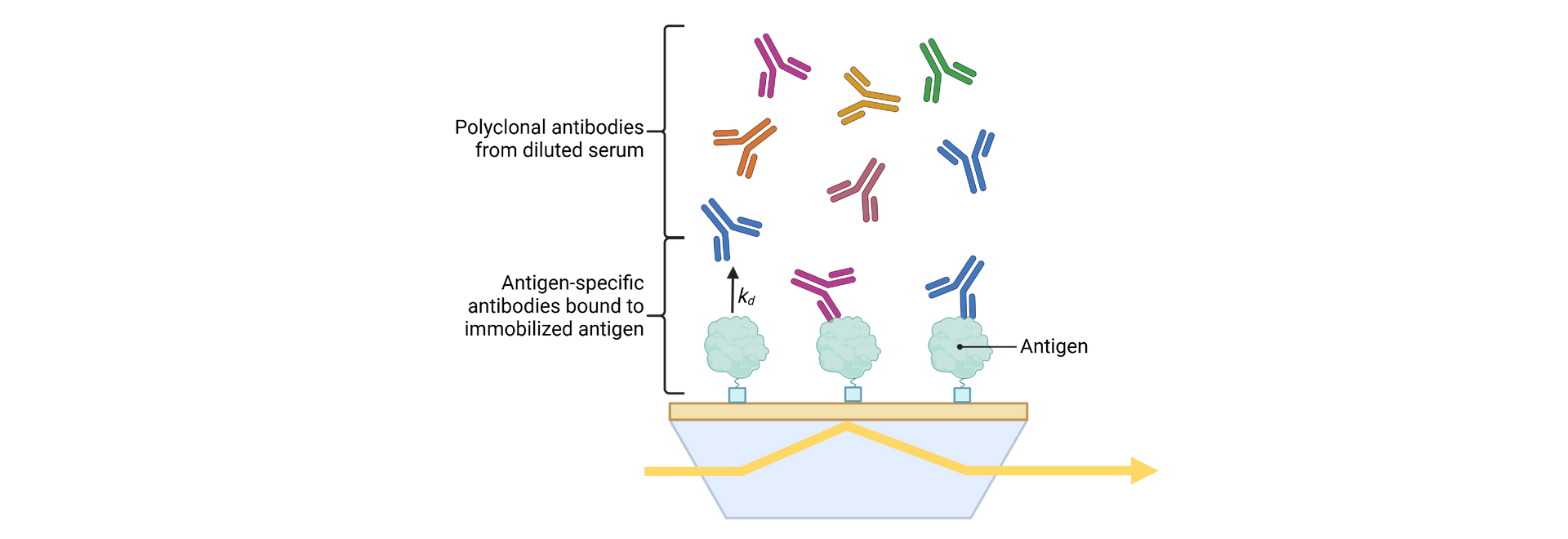
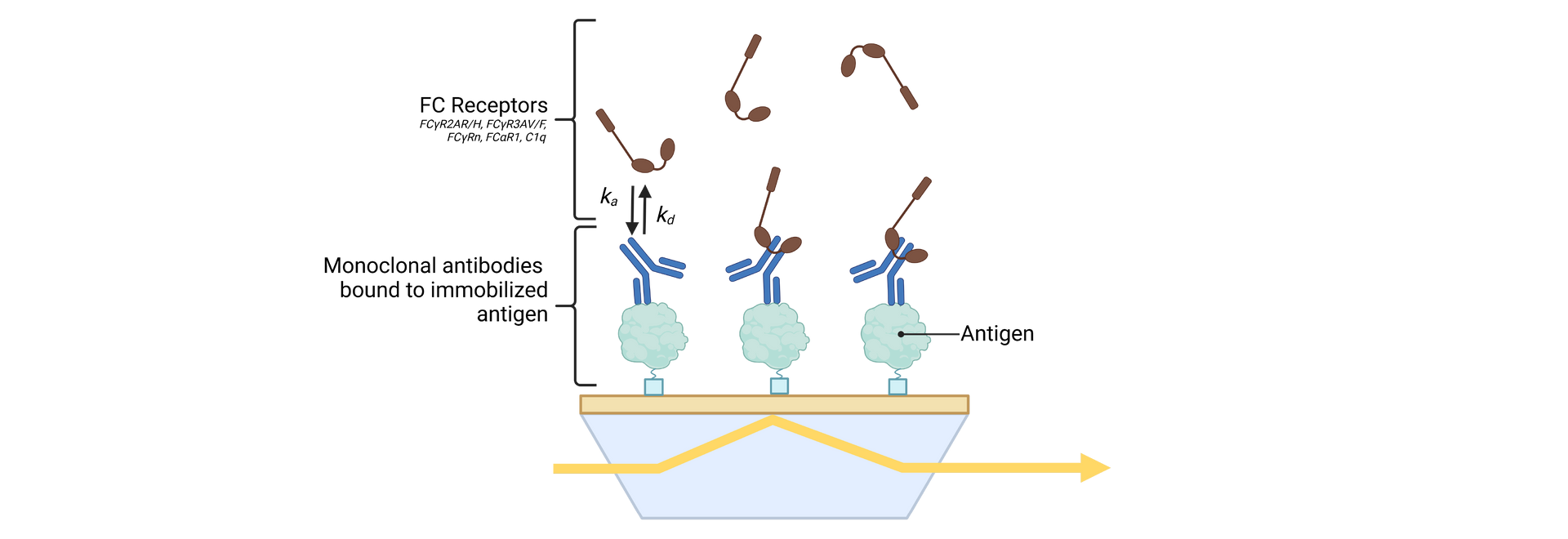
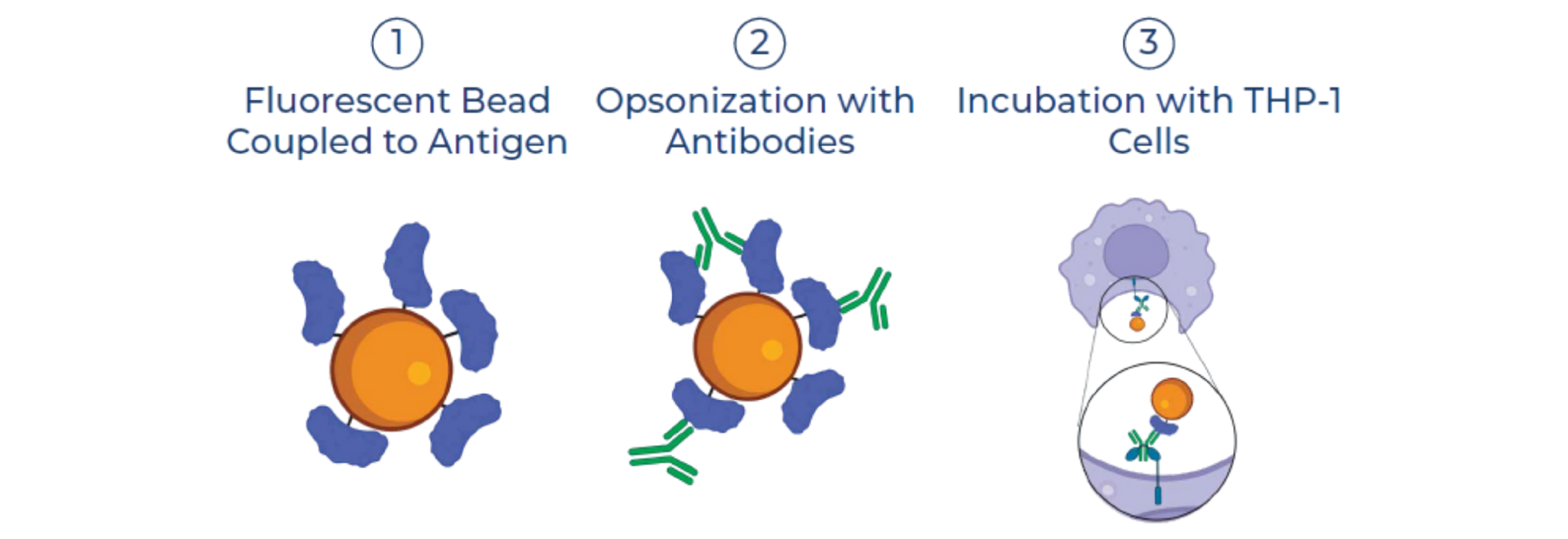
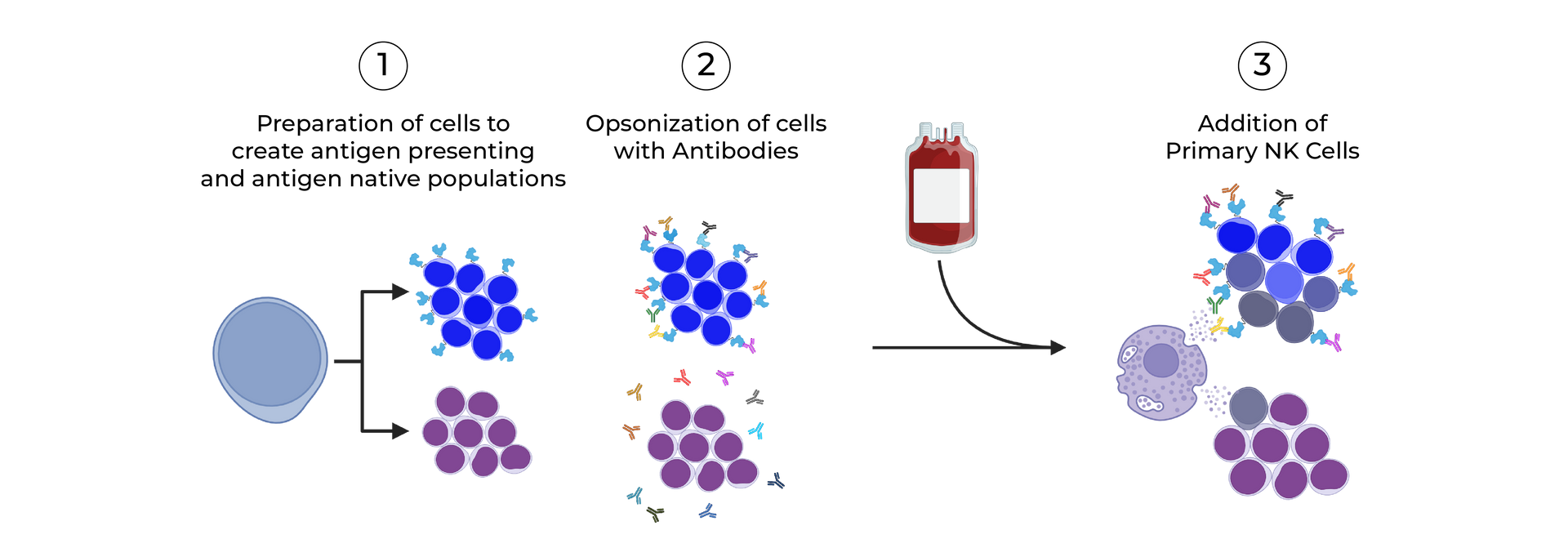
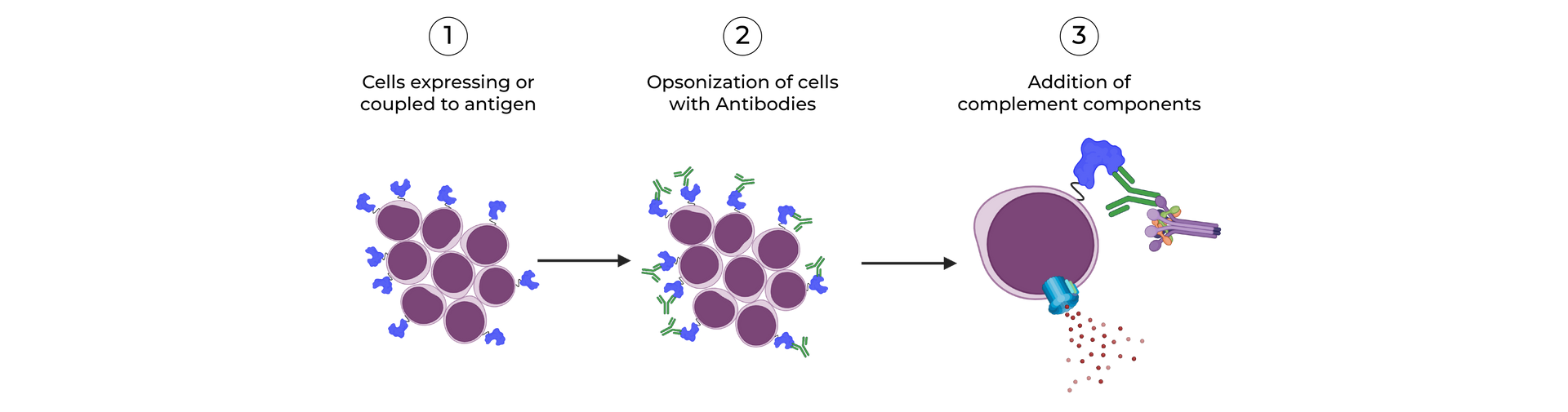
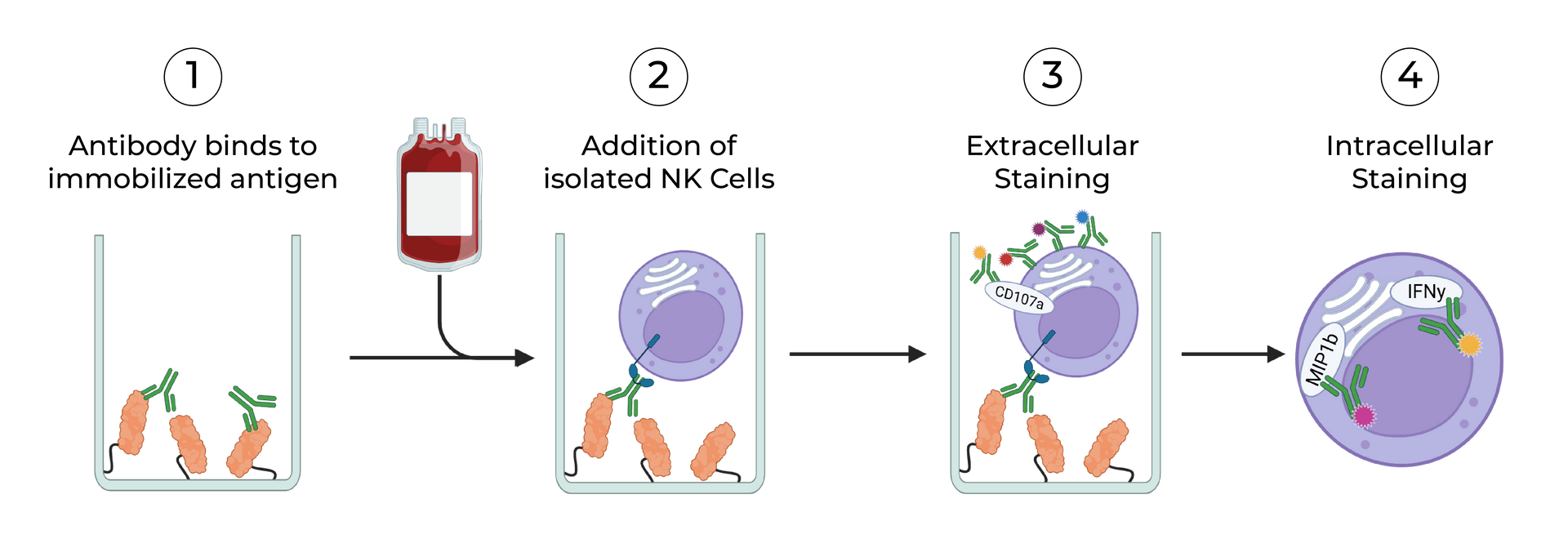
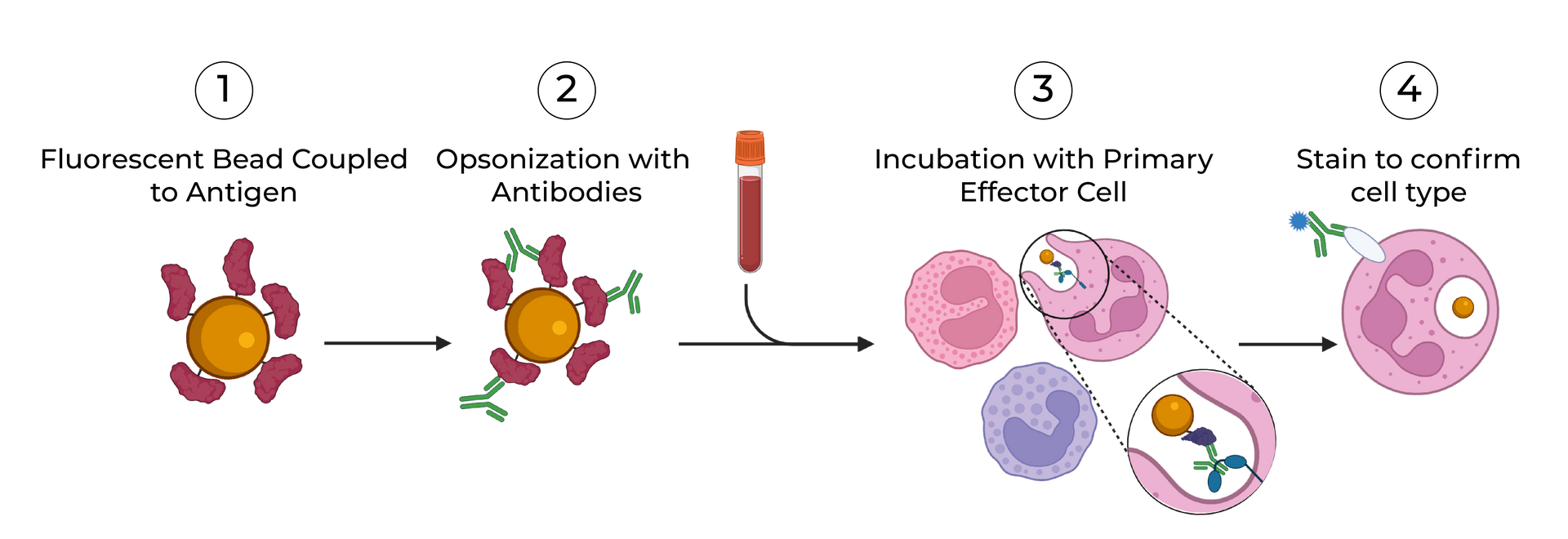
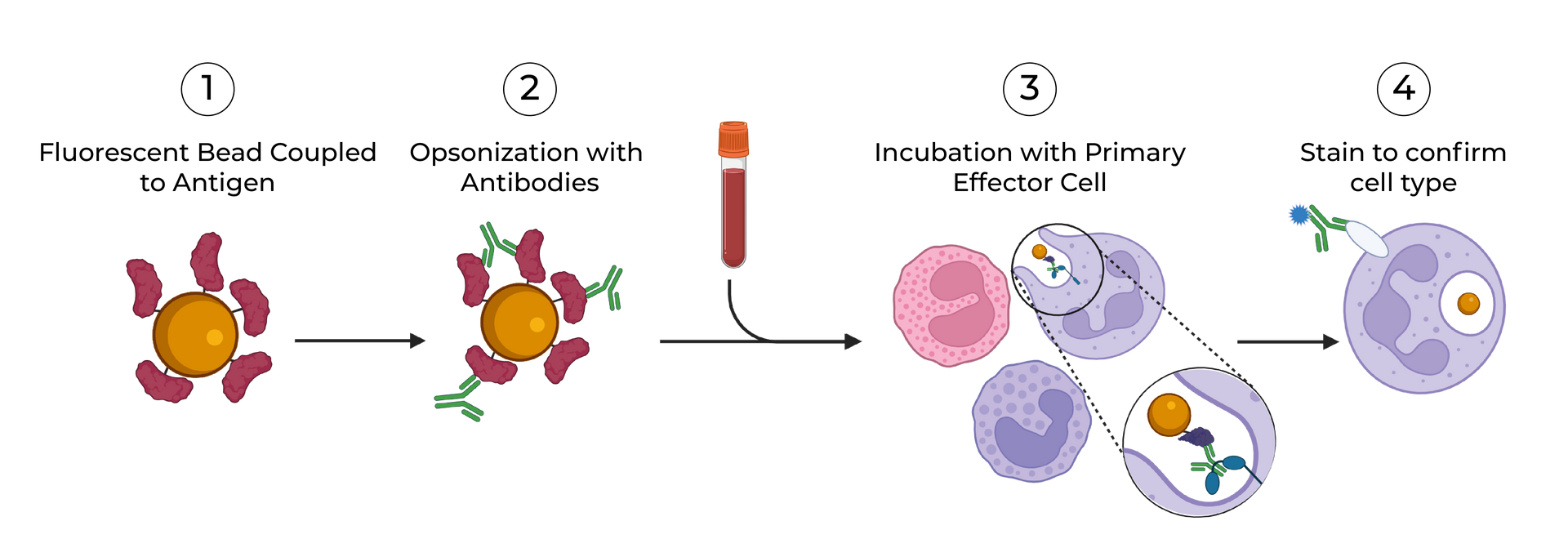
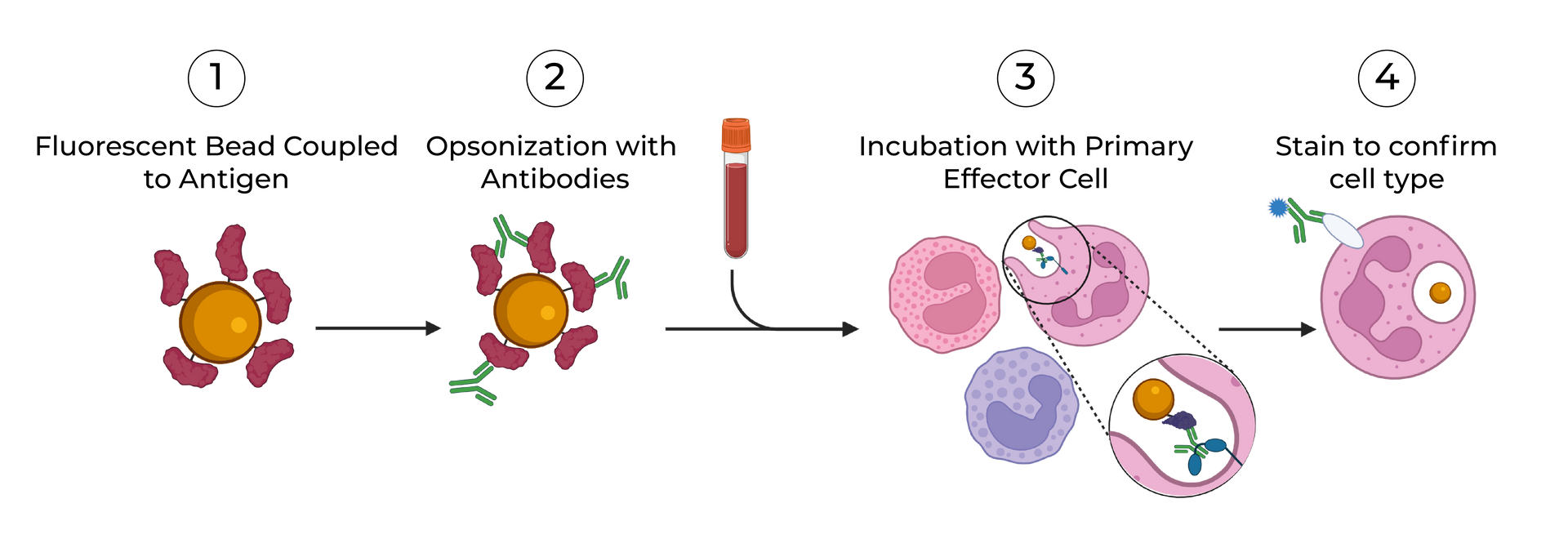
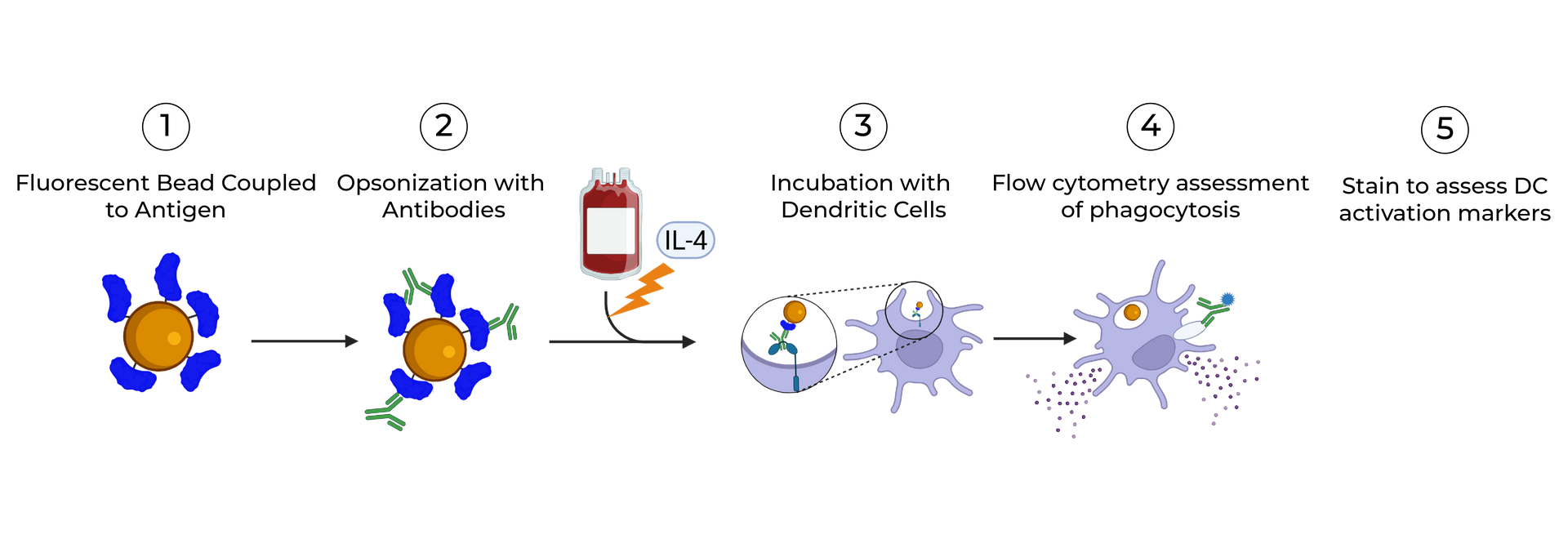
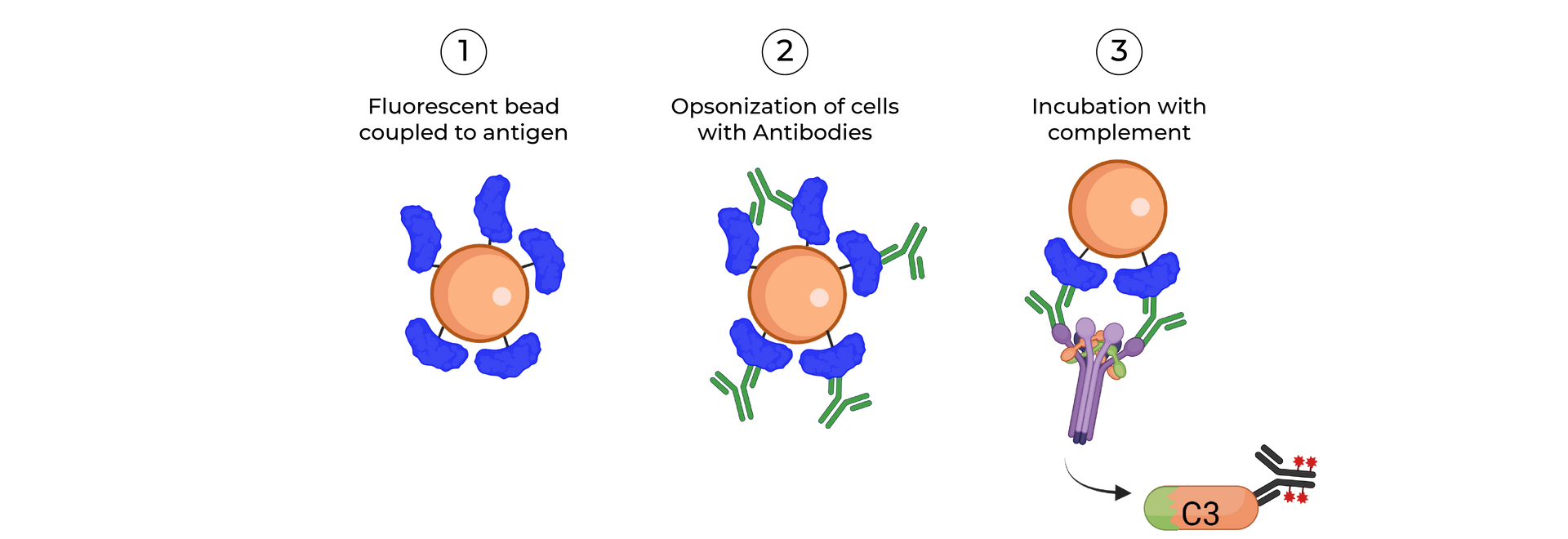
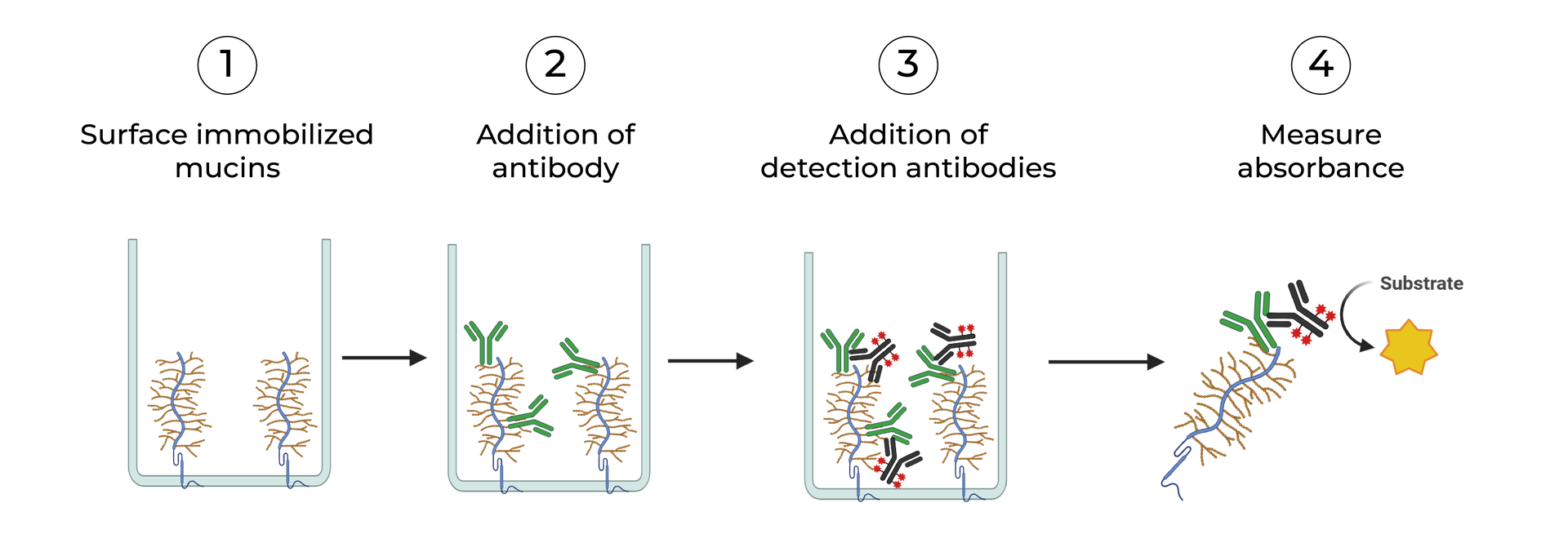
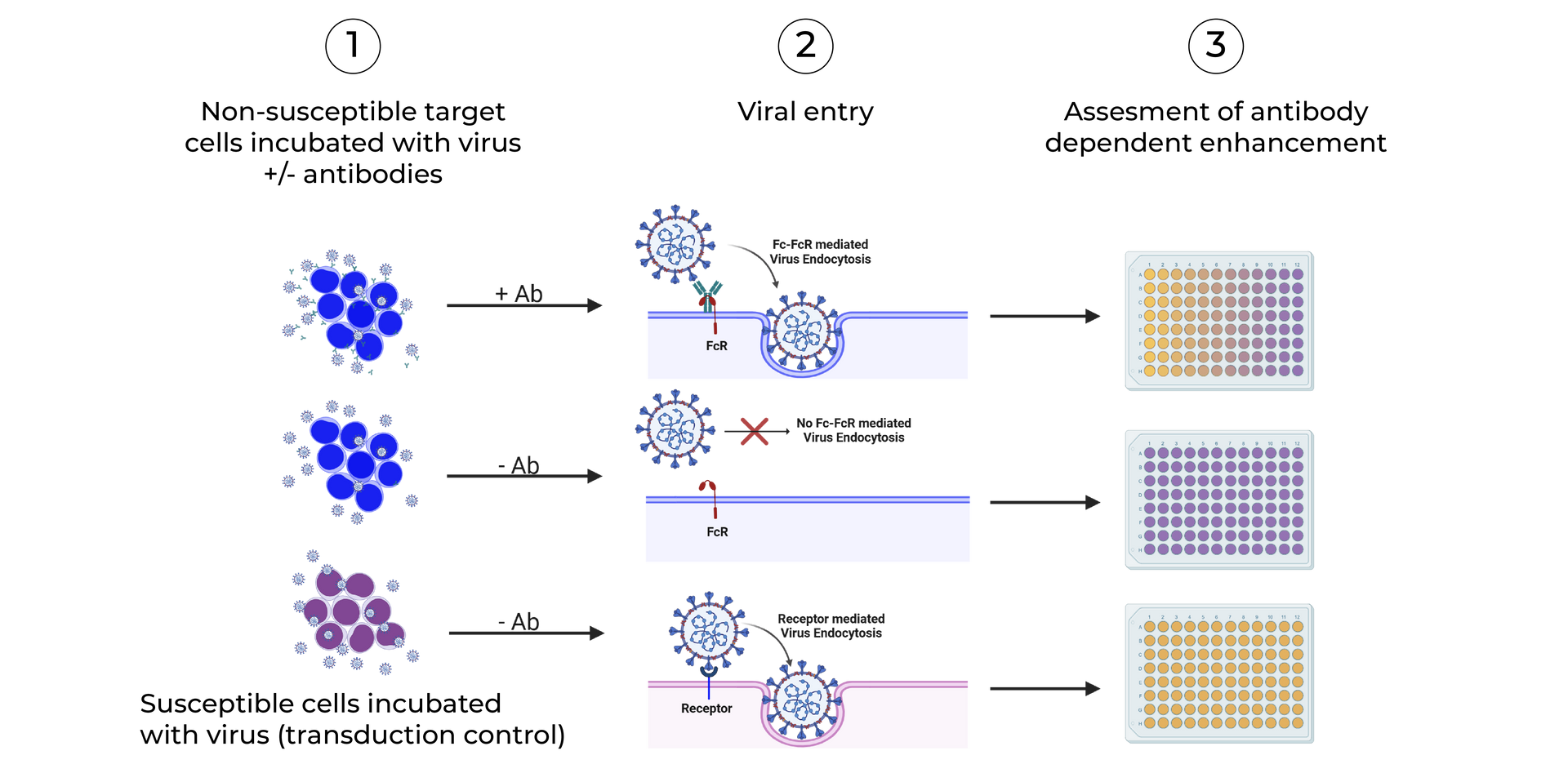
SeromYx combines an extensive suite of biophysical interaction assays and primary cell–based functional assays to characterize the full spectrum of Fc-mediated antibody activity.
Our platform connects Fc structure to immune function, enabling teams to understand mechanism of action (MoA) and make informed development decisions.
Assays are performed in scalable 96–384-well formats and can process thousands of samples per run. We support human, non-human primate (NHP), and murine systems, with additional species available upon request.
BIOPHYSICAL ASSAYS
CELLULAR FUNCTIONAL ASSAYS
Frequently Asked Questions (FAQ)
What is Systems Serology?
Systems Serology is a systems-level immune profiling approach that combines Fc effector function assays, Fc receptor binding measurements, and computational analytics to define how antibodies function, not just what they bind.
How is SeromYx different from other CROs?
Most CROs offer Fc assays as part of a broader service menu.
SeromYx is built entirely around Fc biology.
Our scientific team specializes in understanding how antibody structure, subclass, and receptor engagement translate into functional immune outcomes. That depth allows us to design smarter studies, ask better mechanistic questions, and generate data that meaningfully informs development decisions.
Do you support both vaccines and antibody therapeutics?
Yes. We support both vaccine and antibody therapeutic developers across discovery, preclinical, and clinical stages.
- For vaccines, we help define functional correlates of protection and immune durability.
- For antibody therapeutics, we characterize Fc-driven mechanisms of action, safety signals, and engineering outcomes.