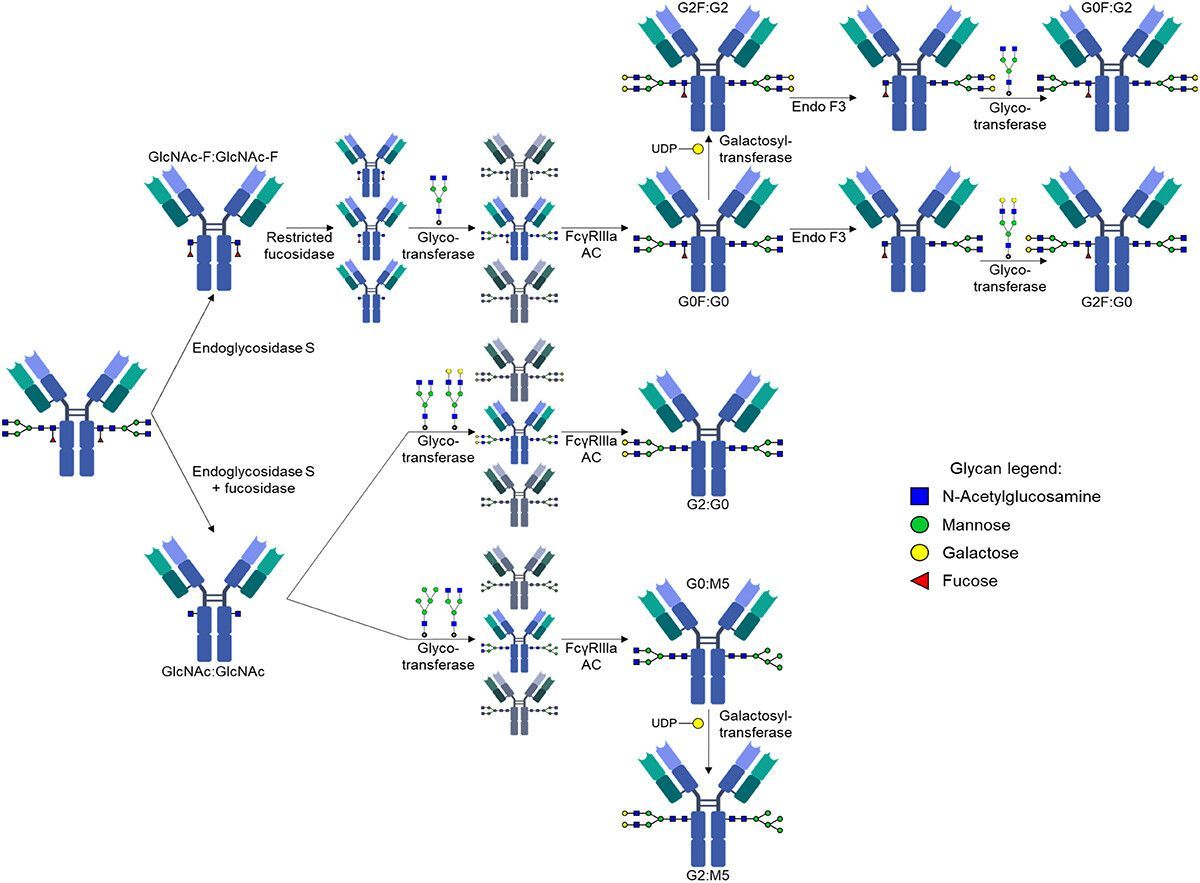
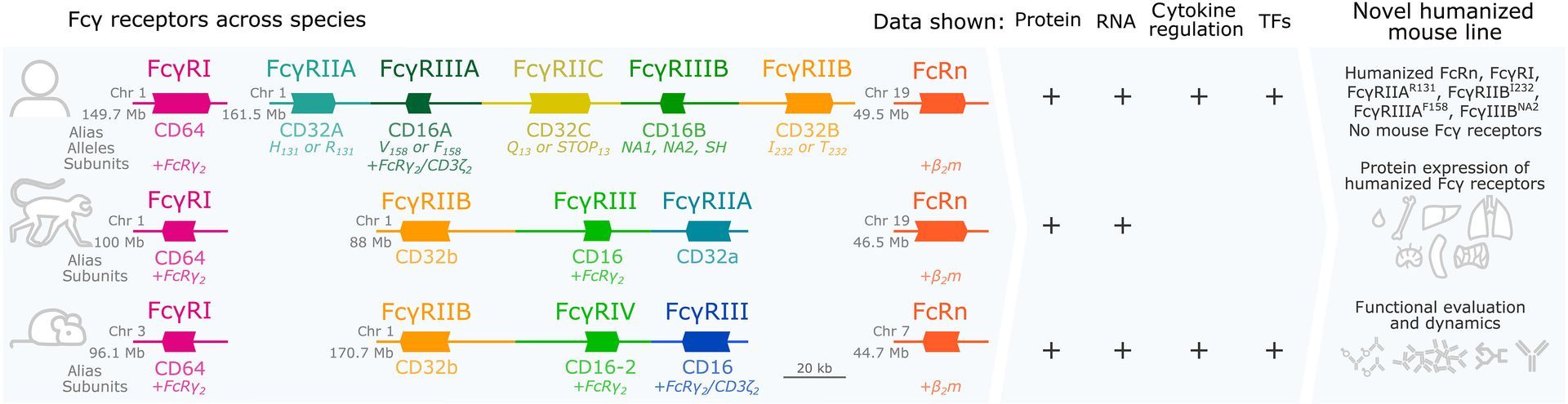
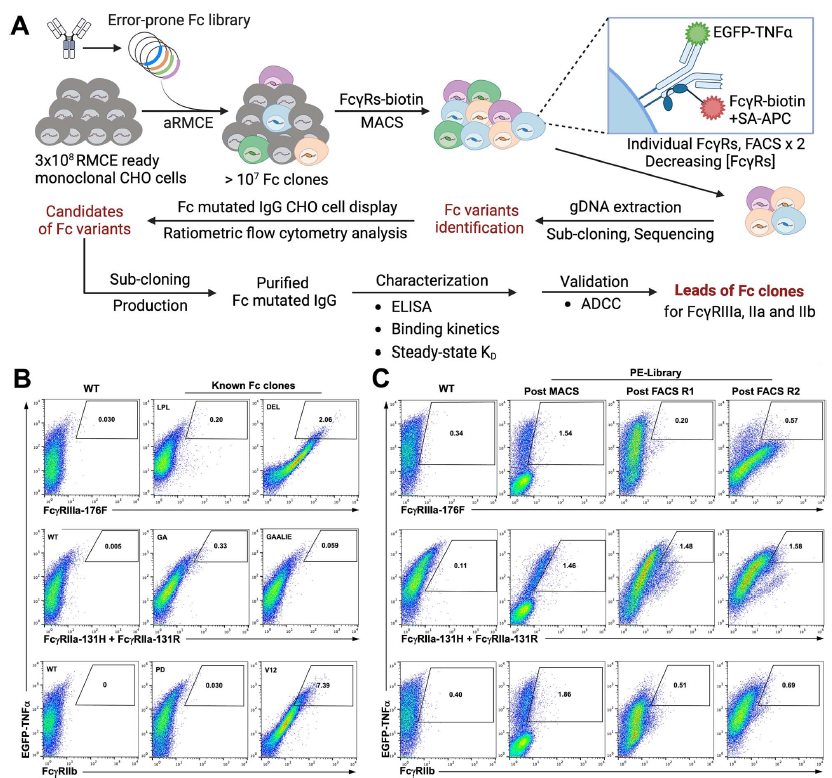
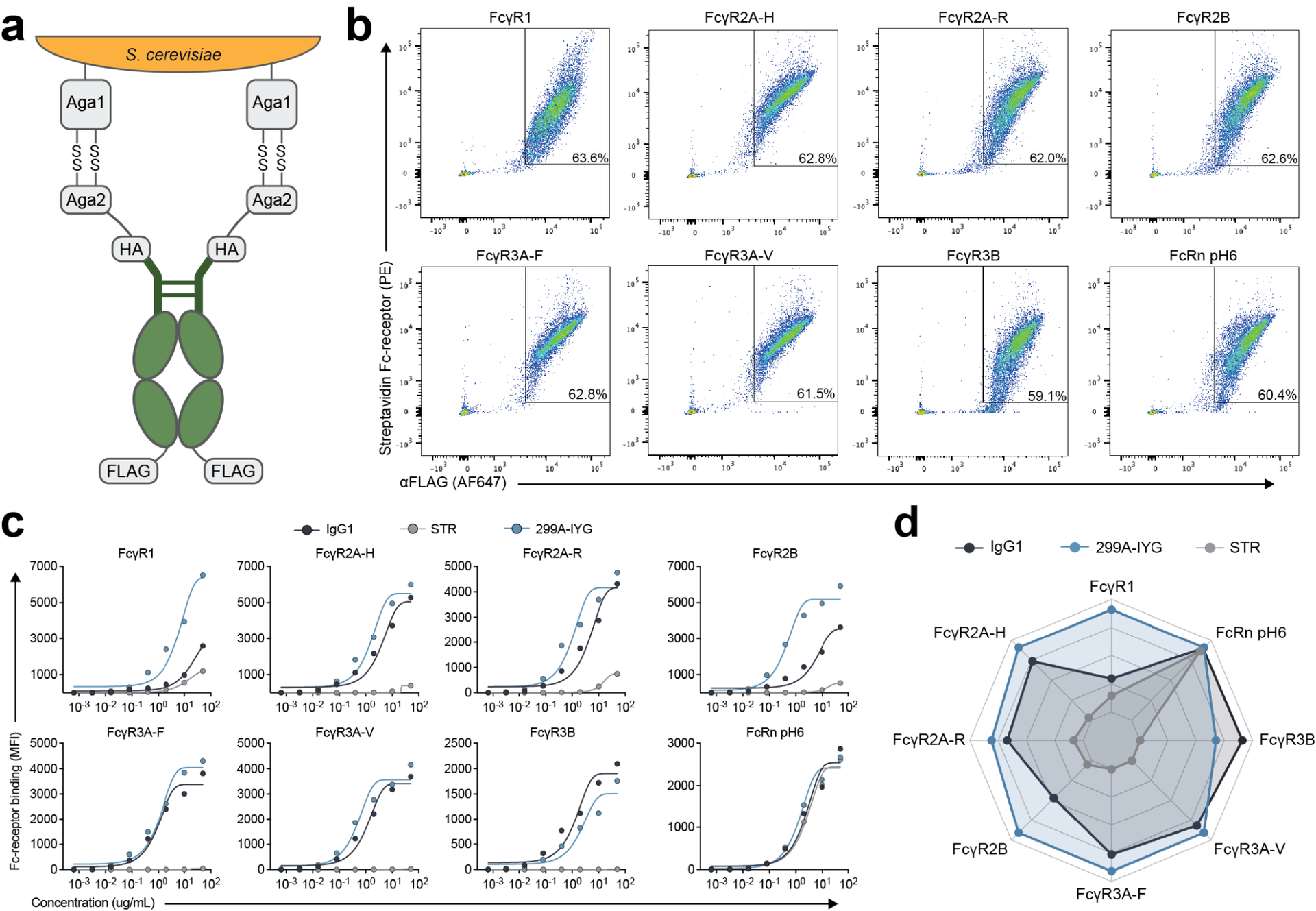
Impacts of Antibody Structure and Mixtures on Receptor Signaling for Antibody-Dependent Cellular Cytotoxicity
The Fc Review:
How do antibody structure and binding dynamics shape ADCC signaling?
A recent study in
The AAPS Journal explores how multiple antibody design features, including Fab affinity, hinge flexibility, Fc engagement, and antigen density, influence antibody-dependent cellular cytotoxicity (ADCC) signaling.
Background:
ADCC is a key mechanism of action for many therapeutic antibodies, yet the strength of this response depends on more than Fcγ receptor binding alone. Productive immune activation requires antibodies to bridge target cells and effector cells within the immune synapse, forming receptor crosslinks that trigger downstream signaling.
Using a combination of computational modeling and experimental data, this study examines how structural properties of antibodies influence these crosslinking events and ultimately shape ADCC signaling outcomes.
The study highlights:
- ADCC signaling is influenced by multiple antibody properties, including Fab affinity, hinge flexibility, Fc receptor engagement, and antigen expression levels.
- Antibody structure affects how efficiently Fc receptors and target antigens are crosslinked within the immune synapse.
- In some cases, moderate Fab affinity supported stronger signaling by favoring productive crosslinking configurations.
- Antibody valency and hinge flexibility also influenced signaling outcomes by altering the geometry of immune synapse interactions.
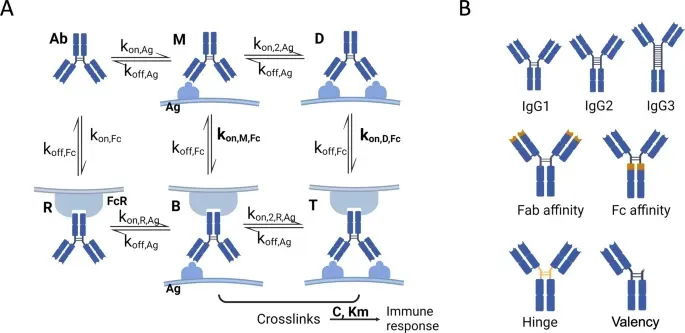
Fig. 1. Proposed kinetic model and antibody variants. A The kinetic model of IgG antibody interactions with an effector cell and an antigen expressing cancer cell. Reversible binding rates determine the interchange between the different states. The states include unbound antibody (Ab), antibody monovalently bound to antigen (M), antibody bivalently bound to two antigens (D), antibody with only the Fc domain bound to the Fc receptor (R), antibody crosslinking one antigen and Fc receptor (B), and a trivalently bound antibody with all three domains bound (T). The two Fab arms of the antibody bind to antigens with on- and off-rates (kon,Ag and koff,Ag). The Fc region engages Fc receptors with corresponding on- and off-rates (kon,Fc and koff,Fc). The antigen-bound antibody can interact with an additional receptor with a fitted effective binding rate (kon,M,Fc and kon,D,Fc). Upon cross-linking of the antibody between the effector cell and cancer cell (states B and T), effector cells are activated for immune induction, which is captured with Michaelis–Menten kinetics by C for the maximum response and Km for cross-linked receptors at half maximum signal. B The varied antibody parameters experimentally tested and modeled in this work include antibody isotype, Fab affinity, Fc affinity, hinge flexibility, and valency (single Fab arm). Figure was created in BioRender.
Implications for antibody development:
For developers designing antibodies with Fc-mediated mechanisms of action, these findings highlight the importance of considering how structural and binding parameters work together to drive effector cell activation.
ADCC potency is not governed by a single variable. Instead, it emerges from the combined effects of antibody affinity, geometry, receptor engagement, and antigen context.
Our perspective:
This study reinforces a key theme in antibody engineering: Fc-mediated function is highly context dependent. Small changes in affinity, hinge architecture, or receptor engagement can reshape how antibodies form immune synapses and activate effector pathways.
Comprehensive Fc profiling helps reveal these effects early, allowing teams to understand how design decisions translate into functional immune responses.
References:
Guan J, Wang T, Tessier PM, Thurber GM. Impacts of Antibody Structure and Mixtures on Receptor Signaling for Antibody-Dependent Cellular Cytotoxicity. The AAPS Journal. 2025;28:39.