Glycan pairing in therapeutic IgG orchestrates Fcγ receptor engagement and ADCC: an integrated structure-function approach for thorough evaluation of Fc N-glycans as critical quality attributes
The Fc Review:
Are we oversimplifying Fc glycosylation?
A recent study in
mAbs explores how glycan pairing within IgG antibodies shapes Fcγ receptor engagement and ADCC, highlighting an often-overlooked layer of Fc biology.
Background:
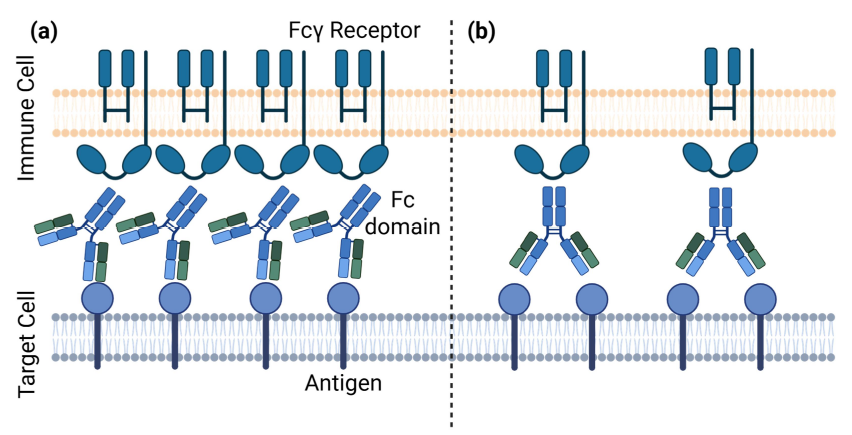
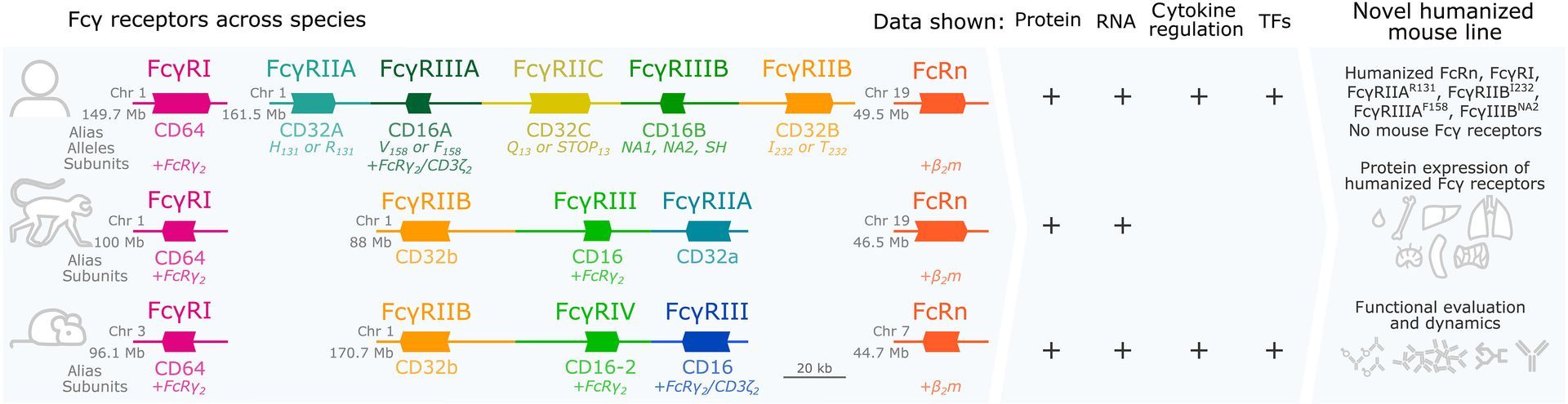
Fc glycosylation is a well-known modulator of antibody function, influencing Fcγ receptor binding and downstream effector activity. However, each IgG contains two Fc glycans, one on each heavy chain, that can pair symmetrically or asymmetrically.
Most analytical approaches report glycan composition as an average, without capturing how these glycans are distributed across individual molecules.
This study asks a key question. Does glycan pairing itself influence Fc function?
The study highlights:
- Fc glycan pairing, not just overall composition, influences Fcγ receptor binding across multiple receptors and allotypes
- A single afucosylated glycan was sufficient to drive enhanced FcγRIIIa binding and ADCC, with limited additional benefit from full afucosylation
- Galactosylation and high-mannose structures showed receptor- and context-dependent effects on Fc engagement
- Functional outcomes cannot be fully inferred from bulk glycan profiles, as different pairing configurations can produce distinct biological behavior
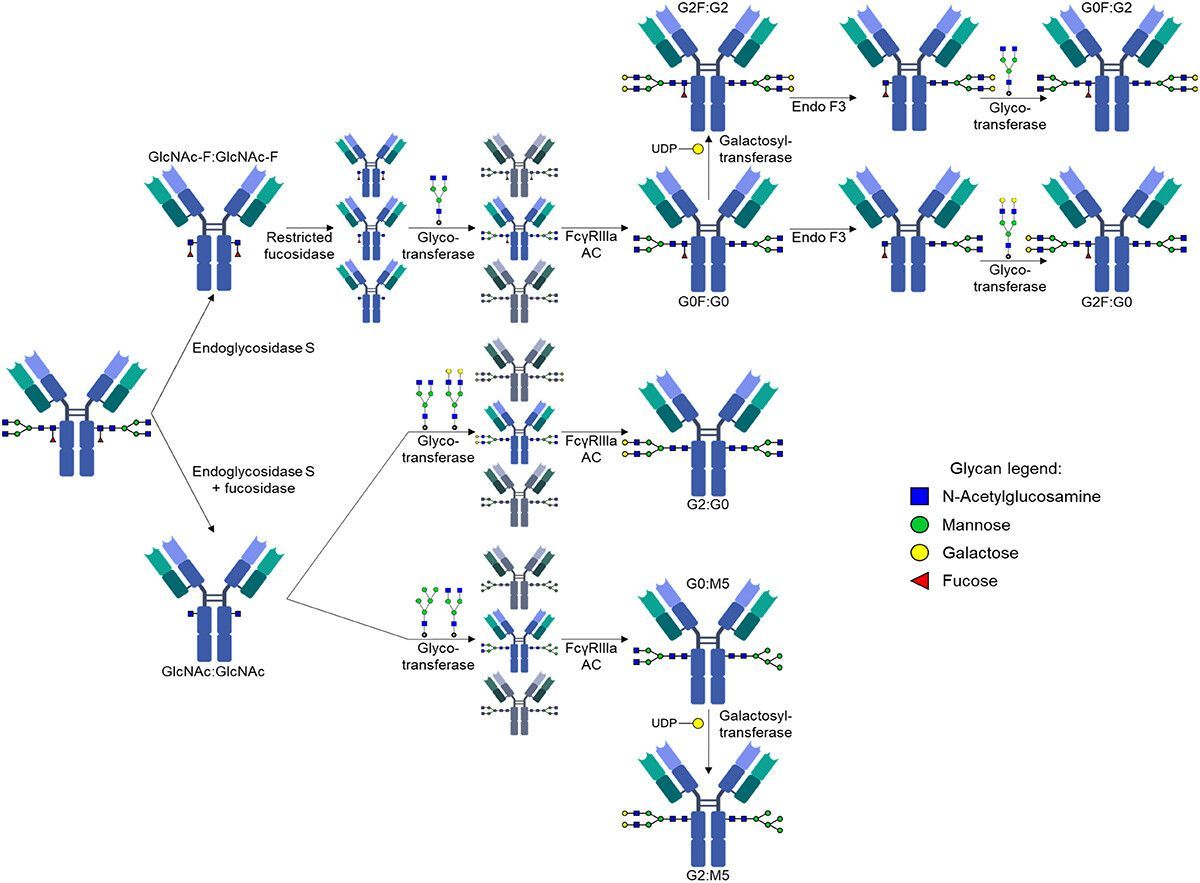
Figure 1. Schematic illustration of the stepwise generation of asymmetrically glycosylated rituximab variants.
Implications for antibody development:
For antibody developers, these findings highlight an important consideration. Fc-mediated function is shaped not only by which glycans are present, but how they are arranged within each molecule.
Relying on averaged glycan measurements may overlook meaningful differences in Fc receptor engagement and effector function.
Our perspective:
This work reinforces a broader theme in antibody engineering. Fc biology is driven by combinations of structural features that work together to shape function.
Even well-characterized modifications such as afucosylation can have nuanced effects depending on how they are incorporated. Evaluating these features in isolation or as averaged properties may miss important aspects of antibody behavior.
Comprehensive Fc profiling helps capture these interactions and provides a clearer view of how design and production choices translate into functional immune responses.
References:
Meudt, M., Baumeister, J., Russell, A. C., Dzielak, L., Hansen, G., Mizaikoff, B., … Higel, F. (2026). Glycan pairing in therapeutic IgG orchestrates Fcγ receptor engagement and ADCC: an integrated structure-function approach for thorough evaluation of Fc N-glycans as critical quality attributes. mAbs, 18(1). https://doi.org/10.1080/19420862.2026.2652642