Antibody Function
The Many Roles of Antibodies Beyond Neutralization
Antibody Function
The Many Roles of Antibodies Beyond Neutralization
Going beyond traditional quantity (titer), binding and neutralization, antigen-specific antibody functionality is mediated via NK cells, Monocytes, Granulocytes and Dendritic cells. The final effectiveness of an antibody derives from a combination of these functions and determining this polyfunctional profile is ideal for successful product development.
Neutralization Activity
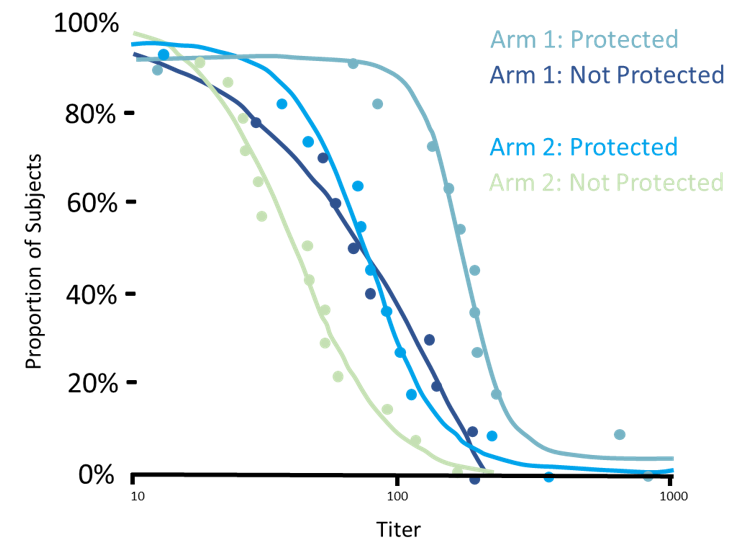
In vaccinology the dogma has been that high titers and neutralizing activity are sufficient for protection. It has become increasingly clear that this dogma is false, especially for difficult vaccine targets because pathogens that are hyper variable (such as HIV), have a complex lifecycle (such as malaria), or have evolved sophisticated immune evasion mechanisms (such as HSV).
The Importance of Immune Effector Functions
Beyond titer and neutralization, antibodies mediate a complex array of mechanisms of immune destruction, of relevance not only to vaccine development but also to immunotherapies in infectious disease, cancer, and autoimmunity.
Developing vaccines to these targets as well as improving existing vaccines which are suboptimal, such as influenza or pertussis, requires much better insight into the functional quality of the antibody produced.
Multiple Factors Modulate Function
Both the specific mechanisms and the degree of function elicited by a given antibody is determined by the interaction between the antibody and Fcγ receptors on innate immune cells and C1q, the recognition molecule of the complement system.
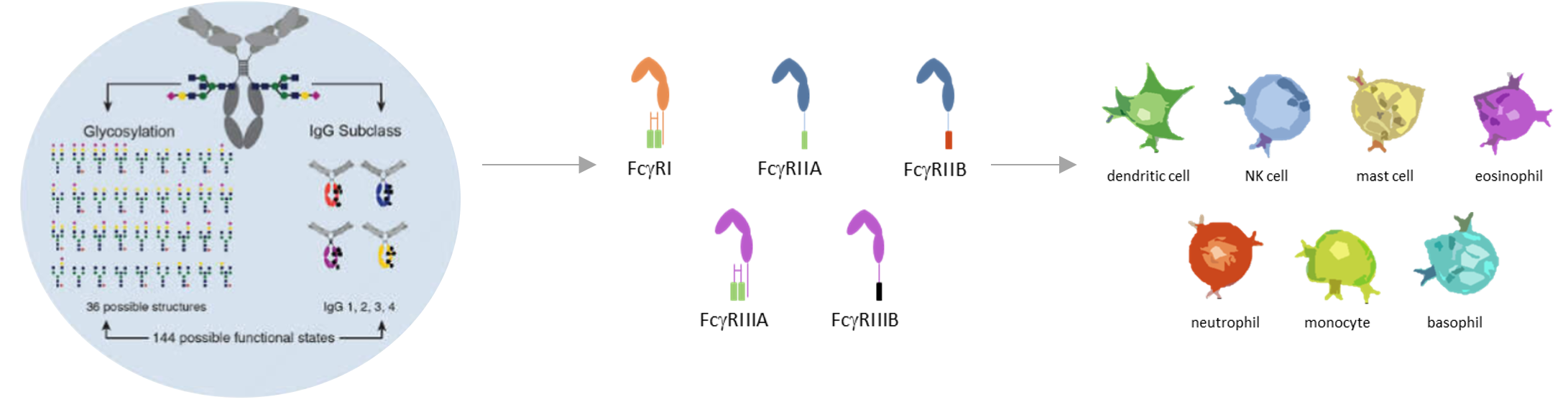
In turn, this is determined by the antibody subclass and isotype and glycan structure of the Fc region of the antibody. Function is also modulated by the specific epitope on the target bound by the antibody.
The importance of immune effector function such as antibody dependent cellular cytotoxicity (ADCC) or antibody-dependent cellular phagocytosis (ADCP) has been well understood by the developers of cancer therapies such as trastuzumab or obinutuzumab, but high throughput tools for screening for desired function have been lacking, until now.
SeromYx platform can measure function in an epitope dependent way, and tie it back to the specific structure of the antibody, shedding a unique insight on this complex interaction.
Functionality is dependent on a complex interplay between the antigen binding and crystallizable fragment domains of an antibody, complement and innate immune cells.
The most basic function of an antibody resides in its antigen-binding F(ab)2 fragments. Paratopes within the Fab bind to epitopes on the surface of target antigen and it is this specific antigen-antibody interaction that results in the fundamental effect: binding to pathogen surfaces and blocking infectivity via disruption of entry proteins and their receptors, neutralizing toxins, neutralizing secreted cellular mediators and affecting molecular pathways, binding cell surface proteins and modulating intra- and inter-cellular activity.
Specific, neutralizing, effects of antibodies are potentiated by extra-neutralizing effects when antibody-antigen immune complexes form. Traditionally, the crystallizable fragment (Fc) domain is considered as a constant region, yet it is here where these variable effects reside, initiated and orchestrated by specific epitope recognition and binding. Variabilities arise from three interrelated biophysical characteristics: isotype and subclass, glycosylation and Fc receptor binding, resulting in potentially 144 different functional states.
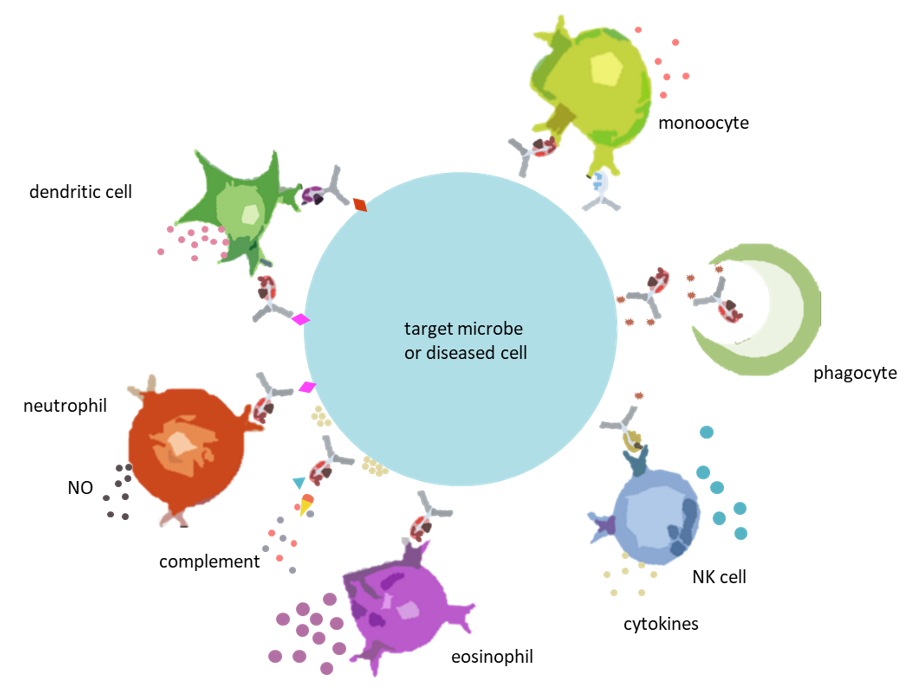
Cells of the innate immune system (monocytes, granulocytes and NK cells) with surface Fc receptors are the final orchestrators of cell mediated effects via activation, secretion of soluble mediators and manifested as cellular lysis and phagocytosis. Solely measuring biophysical characteristics does not clarify cellular functional responses, as epitope binding and Fc mediated effects are also interrelated. Not all epitopes for the same antigen generate the same functional responses. Thus overall, antibody functional responses are influenced by epitope binding and biophysical characteristics.
Additionally, antibodies also mediate effects via activation of the classical complement pathway by oligomerizing and binding C1q, initiating a chain of events that eventually leads to cellular lysis by the formation of a membrane attack complex (MAC).
The importance of immune effector function such as antibody dependent cellular cytotoxicity (ADCC) or antibody-dependent cellular phagocytosis (ADCP) has been well understood by the developers of cancer therapies such as trastuzumab or obinutuzumab, but high throughput tools for screening for desired function have been lacking, until now. SeromYx’s comprehensive suite of antibody functional assays, tying structure to function, in an epitope-dependent manner and integrated with machine learning algorithms, provides the necessary insights to guide successful clinical outcomes for vaccine and immunotherapy products.
Let's Talk - Contact us today to discuss your project.