Case Study
Manufacturing Scale Up
Case Study
Manufacturing Scale Up
Context
Virtually all therapeutic monoclonal antibodies used in humans are produced in mammalian cell lines in vitro. While advances in cell culture have made the manufacturing of monoclonal antibodies more feasible, changes in cell culture conditions or choice of substrate during this scale-up can potentially impact the glycosylation of a monoclonal antibody, which plays a critical role in controlling the functional activity of an antibody, a finding that has been heavily exploited by the therapeutic monoclonal antibody field. In addition to directly monitoring the glycosylation of various production lots of a monoclonal antibody, Systems Serology can be used to evaluate the functional profile of these different lots of antibodies to demonstrate the functional activity of an antibody has not changed due to changes in cell culture conditions that may alter antibody glycosylation.
Problem
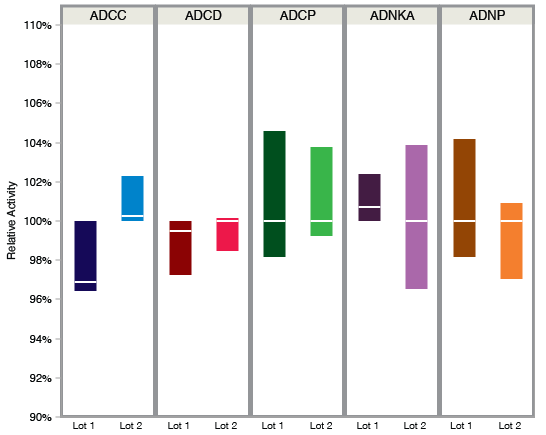
Of the course of the clinical development of a monoclonal antibody, the scale at which the antibody needs to be produced dramatically increases from the micrograms needed during early pre-clinical development to the hundreds of grams of antibody needed for a phase II clinical trial. With this large increase in the amount of antibody needed, the approach to antibody production often changes from in-house, small-batch, transient production of antibodies during pre-clinical development to out-sourced, large-scale, bio-reactor production as clinical development progresses. These changes in antibody production often include changes in the production cell line (e.g., CHO vs. 293), cell culture conditions (e.g., dissolved oxygen, pH, temperature), and media/feed formulations (e.g., chemical supplements), all of which can significantly impact the glycosylation of the produced antibody. As relatively small changes in antibody glycosylation can have a significant impact on antibody function, the functional activity of antibodies that were selected for particular functional profiles during pre-clinical development must be re-evaluated to confirm that changes in production methodologies did not alter the targeted antibody functions. Here, Systems Serology was used to evaluate the functional activity of a panel of SARS CoV2–specific monoclonal antibodies following scale-up of production.
Systems Serology Application
After switching from in-house antibody production to outsourced, batch-fed antibody production, differences in antibody glycosylation were readily observed, including a decrease in antibody sialylation, a decrease in antibody galactosylation, and an increase in antibody fucosylation. As these antibodies had been previously selected for a specific functional profile, the newly produced antibodies were compared to the original antibodies to confirm that the changes in antibody glycosylation did not alter the functional profile of the antibodies. While lot-to-lot variability was observed in binding to specific Fc receptors within the individual antibody sets, these differences were not large enough to have an impact on the functional activity of the different production runs, as all production runs for a given antibody demonstrated similar functional activities. Therefore, despite the changes in antibody glycosylation associated with changes in antibody production, all antibody production runs had similar functional profiles, demonstrating that the new production methodology did not impact that functional activity of the antibodies.
Conclusion
Using Systems Serology, the functional activity of a panel of SARS CoV2–specific monoclonal antibodies were systematically profiled before and after a change in antibody production methods. While the change to batch-fed production resulted in changes in antibody glycosylation, these changes did not impact the functional profile of the tested antibodies. As monoclonal antibodies that are destined for clinical use are frequently optimized to mediate specific effector functions, the Systems Serology suite of assays offered by SeromYx can be used to comprehensive measure the functional activity of antibodies at all stages of clinical development, ultimately demonstrating that the target functional profile has not changed due to difference in antibody production methodologies.